Antibiotic treatments becoming less effective
In 1979, Bobbie Smith’s four year-old son Chris received a prescription from a general pediatrician. After two rounds of antibiotic treatment for what seemed to be a routine infection, Chris developed diarrhea and had a hard time waking up. His mother took him to a specialist.
“When I parked in underground parking, a bunch of doctors came out to help me carry Chris in, it was like a movie,” Smith says, who asked to go by her maiden name. “They were very excited because this was a test of a new theory.”
The four year-old had contracted a C. diff infection, a bacterial disease that invades the intestines and causes lesions to form. C. diff had been recognized and studied for years leading up to Chris’s infection. But the treatment Chris originally received had made the bacteria more robust, and resistant to common antibiotics.
Nearly 40 years after Chris’s infection, the Centers for Disease Control released its first ever report on Antibiotic Resistance Threats in the United States last spring. In it, antibiotic resistant infections (ABIs) are reported to develop in over two million Americans and kill at least 23,000 people each year, with C. diff claiming more than half of those deaths. The report stresses the “potentially catastrophic consequences of inaction” healthcare institutions face with these new bacterial strains. Misuse of antibiotics and highly adaptable bacteria now require researchers, health organizations, and healthcare facilities to follow and react to resistance more than ever before.
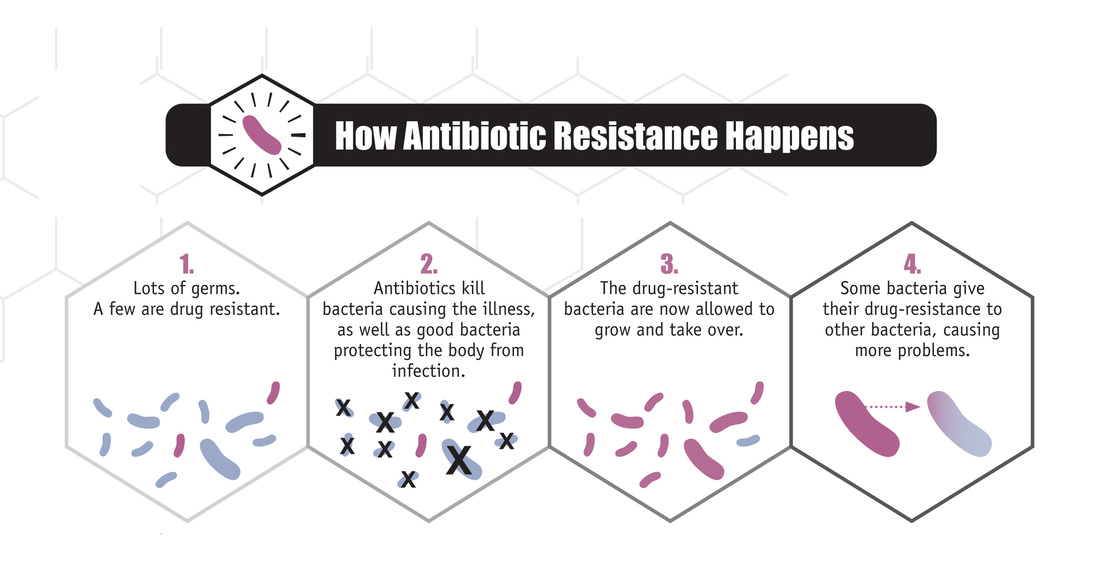
Less than a decade after penicillin was first mass produced in the 1940s, doctors and nurses observed that some infections were less sensitive to antibiotic treatment. The phenomenon, now known as antibiotic resistance, occurs when a bacteria develops a mutation that allows it to degrade the drug it encounters, or to change its genetic makeup so that the drug no longer targets it. While penicillin is still used for some minor infections, it’s more likely to be seen in a museum than in a pill bottle due to the rapid adaptation of some bacteria. The CDC even recommends against using the famous antibiotic in cases of serious infection.
“When I parked in underground parking, a bunch of doctors came out to help me carry Chris in, it was like a movie,” Smith says, who asked to go by her maiden name. “They were very excited because this was a test of a new theory.”
The four year-old had contracted a C. diff infection, a bacterial disease that invades the intestines and causes lesions to form. C. diff had been recognized and studied for years leading up to Chris’s infection. But the treatment Chris originally received had made the bacteria more robust, and resistant to common antibiotics.
Nearly 40 years after Chris’s infection, the Centers for Disease Control released its first ever report on Antibiotic Resistance Threats in the United States last spring. In it, antibiotic resistant infections (ABIs) are reported to develop in over two million Americans and kill at least 23,000 people each year, with C. diff claiming more than half of those deaths. The report stresses the “potentially catastrophic consequences of inaction” healthcare institutions face with these new bacterial strains. Misuse of antibiotics and highly adaptable bacteria now require researchers, health organizations, and healthcare facilities to follow and react to resistance more than ever before.
Less than a decade after penicillin was first mass produced in the 1940s, doctors and nurses observed that some infections were less sensitive to antibiotic treatment. The phenomenon, now known as antibiotic resistance, occurs when a bacteria develops a mutation that allows it to degrade the drug it encounters, or to change its genetic makeup so that the drug no longer targets it. While penicillin is still used for some minor infections, it’s more likely to be seen in a museum than in a pill bottle due to the rapid adaptation of some bacteria. The CDC even recommends against using the famous antibiotic in cases of serious infection.
Even some new antibiotics cannot stop some bacteria from reproducing and spreading says Emory School of Medicine professor Shonna McBride, who studies the bacteria that causes C. diff infection, Clostridium difficile.
C. diff is able to tolerate high concentrations of antibiotics that do not specifically target the bacteria. Unsupervised antibiotic use can harm the useful bacteria in intestines and increase susceptibility to developing resistance. Even with targeted treatment, C. diff can still spread by forming spores, which are “like becoming a seed,” says McBride. “And then they can’t be killed, even with the antibiotics that do target them.”
However, thoroughly washing with regular soap and water can disinfect hands and prevent spread. Bleach and other oxidizing agents can also be used to sterilize surfaces or instruments suspected to contain C. diff and its spores.
The most concerning aspect of these infections is that many of them occur in hospitals, where antibiotic use is most prevalent and disinfecting procedures are most strict. Phuong Nguyen, a nurse at North DeKalb Medical Center, says at least one of her surgery patients on any given day will be isolated from contact because they contracted an antibiotic-resistant infection while in care. Nguyen says that patients are sometimes not given enough time for the antibiotics to take effect, since surgeons want to avoid rescheduling expensive operations. Caring for patients with ABIs requires more expensive antibiotics and can delay treatment. Tufts University estimates that this extra care costs patients and institutions $20 billion annually.
Patients who undergo surgery are not the only high-risk group. Anyone with a compromised immune system is susceptible to contracting a potentially fatal infection. Doctors inform chemotherapy and transplant patients about these risks. Patients of autoimmune diseases such as rheumatoid arthritis are also at high risk of developing resistance.
While antibiotic resistance is already a widespread problem for doctors and nurses, the demands of some patients exacerbate the issue. Many people expect a simple pill to cure them of all disease. With more people receiving healthcare insurance under the Patient Protection and Affordable Care Act, it’s now cheaper for patients to afford prescription drugs.
“When they leave and have their diagnosis done, and there’s no new medical prescriptions, then they’re always complaining about how they need some medication,” says Vincent Zhang, a medical scribe who observes and documents patient-physician interactions at Eastside Medical's Emergency Room. “That’s when the doctor, out of guilt, gives them antibiotics. They are over-prescribing antibiotics to all their patients right now.”
Even after the prescription is written without antibiotics listed, many patients still try to acquire antibiotics under the assumption that more treatment is better. CVS Pharmacy technician Angela Cheng says some patients come in and say “oh, didn’t my doctor write a prescription for antibiotics?” Cheng says standard procedure is to inquire further about their condition and she often discovers the patient would not benefit from antibiotic treatment. “We tell them that they don’t need antibiotics for a viral infection,” Cheng says.
The CDC’s 2013 report proposes some measures that both institutions and patients can take to reduce risk of contracting and spreading ABIs. Medical professionals are urged to “use antibiotics only when they are necessary to treat, and in some cases prevent, disease.” As for patients, the report reiterates two major points all of interviewed medical professionals emphasized.
Nguyen also says that patients who are new to healthcare need to understand that receiving treatment is not like a day at the salon. “You have to put in some work,” she says. And when a patient is discharged, they are not fully cured or recovered. “We manage their symptoms, we teach patients how to manage their symptoms, know about their medications, and it’s an adjustment,” Nguyen says about her patients. “They have to change their lifestyle if they want to keep on going.” However, not all the burden of action falls upon patients and the healthcare system.
Drug resistance is commonly framed as a human disease, but emerging research indicates that antibiotics used when raising livestock is also contributing to ABIs. In the 2011 Report on Antimicrobials Sold or Distributed for Use in Food-Producing Animals, the FDA observed the majority of antibiotics sold in the United States are administered to cows, chickens, pigs and other livestock intended for consumption. Cheng speculates that these pharmaceutical companies can only benefit from increased sales of antibiotics and that development of ABIs allows pharmaceutical manufacturers to sell more novel drugs and at higher prices.
While the European Food Safety Authority has banned the use of many antibiotics in cultivating livestock, the FDA has only implemented a voluntary plan to reduce the inappropriate and unapproved use of antibiotics to increase animal growth.
With a concerted effort from all the parties that use antibiotics, future outbreaks of ABI can be controlled and fatalities can be minimized, says the CDC. The CDC will release an updated report on Antibiotic Resistance Threats in the United States every five years. Between now and 2018, the CDC proposes to improve outdated detection methods, and introduce technologies to predict and prevent outbreaks of ABIs.
The President’s 2014 Budget proposes an Advanced Molecular Detection and Response to Infection Disease Outbreaks initiative, which the World Health Organization (WHO) projects can halve the number of ABI cases compared to traditional detection methods. Hospitals and clinics are expected to submit records of infection, antibiotic use, and resistance to the National Health Care Safety Network. This electronic database is intended to provide researchers with enough statistics to identify markers which would indicate potential ABI outbreak.
But modern medical science is not yet able to completely cure patients of ABIs. One in five patients who have been treated for C. diff experience recurrence of the infection, according to the Mayo Clinic.
Bobbie Smith, now 72, also contracted the same infection that affected her son. She suffered from C. diff from 1993 to 1997 and again in 1999 and 2011. Smith says her bouts with C. diff feel like having a flu for years at a time. In her 60s, Smith was diagnosed with early on-set osteoporosis due to the effect C. diff has on intestinal function. She continues to suffer from frequent pneumonia, which even caused her to reschedule her interview. C. diff has left Smith susceptible to other infections which require antibiotic treatment, antibiotics which have triggered her recurrences in the past.
For nearly 15 years, she has moderated an Internet forum at cdiffsupport.com to help patients learn about and cope with the disease. Smith now advocates before political bodies for improved infectious disease diagnostic procedures and more stringent antibiotic testing, which may help prevent the kind of treatment that developed resistant bacteria in both her and her son.
C. diff is able to tolerate high concentrations of antibiotics that do not specifically target the bacteria. Unsupervised antibiotic use can harm the useful bacteria in intestines and increase susceptibility to developing resistance. Even with targeted treatment, C. diff can still spread by forming spores, which are “like becoming a seed,” says McBride. “And then they can’t be killed, even with the antibiotics that do target them.”
However, thoroughly washing with regular soap and water can disinfect hands and prevent spread. Bleach and other oxidizing agents can also be used to sterilize surfaces or instruments suspected to contain C. diff and its spores.
The most concerning aspect of these infections is that many of them occur in hospitals, where antibiotic use is most prevalent and disinfecting procedures are most strict. Phuong Nguyen, a nurse at North DeKalb Medical Center, says at least one of her surgery patients on any given day will be isolated from contact because they contracted an antibiotic-resistant infection while in care. Nguyen says that patients are sometimes not given enough time for the antibiotics to take effect, since surgeons want to avoid rescheduling expensive operations. Caring for patients with ABIs requires more expensive antibiotics and can delay treatment. Tufts University estimates that this extra care costs patients and institutions $20 billion annually.
Patients who undergo surgery are not the only high-risk group. Anyone with a compromised immune system is susceptible to contracting a potentially fatal infection. Doctors inform chemotherapy and transplant patients about these risks. Patients of autoimmune diseases such as rheumatoid arthritis are also at high risk of developing resistance.
While antibiotic resistance is already a widespread problem for doctors and nurses, the demands of some patients exacerbate the issue. Many people expect a simple pill to cure them of all disease. With more people receiving healthcare insurance under the Patient Protection and Affordable Care Act, it’s now cheaper for patients to afford prescription drugs.
“When they leave and have their diagnosis done, and there’s no new medical prescriptions, then they’re always complaining about how they need some medication,” says Vincent Zhang, a medical scribe who observes and documents patient-physician interactions at Eastside Medical's Emergency Room. “That’s when the doctor, out of guilt, gives them antibiotics. They are over-prescribing antibiotics to all their patients right now.”
Even after the prescription is written without antibiotics listed, many patients still try to acquire antibiotics under the assumption that more treatment is better. CVS Pharmacy technician Angela Cheng says some patients come in and say “oh, didn’t my doctor write a prescription for antibiotics?” Cheng says standard procedure is to inquire further about their condition and she often discovers the patient would not benefit from antibiotic treatment. “We tell them that they don’t need antibiotics for a viral infection,” Cheng says.
The CDC’s 2013 report proposes some measures that both institutions and patients can take to reduce risk of contracting and spreading ABIs. Medical professionals are urged to “use antibiotics only when they are necessary to treat, and in some cases prevent, disease.” As for patients, the report reiterates two major points all of interviewed medical professionals emphasized.
- If prescribed antibiotics, take them exactly as directed. Do not cut short your regimen after your symptoms have disappeared. Doing so increases the chance of developing antibiotic resistance.
- Thoroughly wash your hands after you interact with physicians or nurses. Wash your hands for at least 15 seconds and do so frequently, especially after diarrhea (which may be an indicator of C. diff infection).
Nguyen also says that patients who are new to healthcare need to understand that receiving treatment is not like a day at the salon. “You have to put in some work,” she says. And when a patient is discharged, they are not fully cured or recovered. “We manage their symptoms, we teach patients how to manage their symptoms, know about their medications, and it’s an adjustment,” Nguyen says about her patients. “They have to change their lifestyle if they want to keep on going.” However, not all the burden of action falls upon patients and the healthcare system.
Drug resistance is commonly framed as a human disease, but emerging research indicates that antibiotics used when raising livestock is also contributing to ABIs. In the 2011 Report on Antimicrobials Sold or Distributed for Use in Food-Producing Animals, the FDA observed the majority of antibiotics sold in the United States are administered to cows, chickens, pigs and other livestock intended for consumption. Cheng speculates that these pharmaceutical companies can only benefit from increased sales of antibiotics and that development of ABIs allows pharmaceutical manufacturers to sell more novel drugs and at higher prices.
While the European Food Safety Authority has banned the use of many antibiotics in cultivating livestock, the FDA has only implemented a voluntary plan to reduce the inappropriate and unapproved use of antibiotics to increase animal growth.
With a concerted effort from all the parties that use antibiotics, future outbreaks of ABI can be controlled and fatalities can be minimized, says the CDC. The CDC will release an updated report on Antibiotic Resistance Threats in the United States every five years. Between now and 2018, the CDC proposes to improve outdated detection methods, and introduce technologies to predict and prevent outbreaks of ABIs.
The President’s 2014 Budget proposes an Advanced Molecular Detection and Response to Infection Disease Outbreaks initiative, which the World Health Organization (WHO) projects can halve the number of ABI cases compared to traditional detection methods. Hospitals and clinics are expected to submit records of infection, antibiotic use, and resistance to the National Health Care Safety Network. This electronic database is intended to provide researchers with enough statistics to identify markers which would indicate potential ABI outbreak.
But modern medical science is not yet able to completely cure patients of ABIs. One in five patients who have been treated for C. diff experience recurrence of the infection, according to the Mayo Clinic.
Bobbie Smith, now 72, also contracted the same infection that affected her son. She suffered from C. diff from 1993 to 1997 and again in 1999 and 2011. Smith says her bouts with C. diff feel like having a flu for years at a time. In her 60s, Smith was diagnosed with early on-set osteoporosis due to the effect C. diff has on intestinal function. She continues to suffer from frequent pneumonia, which even caused her to reschedule her interview. C. diff has left Smith susceptible to other infections which require antibiotic treatment, antibiotics which have triggered her recurrences in the past.
For nearly 15 years, she has moderated an Internet forum at cdiffsupport.com to help patients learn about and cope with the disease. Smith now advocates before political bodies for improved infectious disease diagnostic procedures and more stringent antibiotic testing, which may help prevent the kind of treatment that developed resistant bacteria in both her and her son.
Comment Form is loading comments...